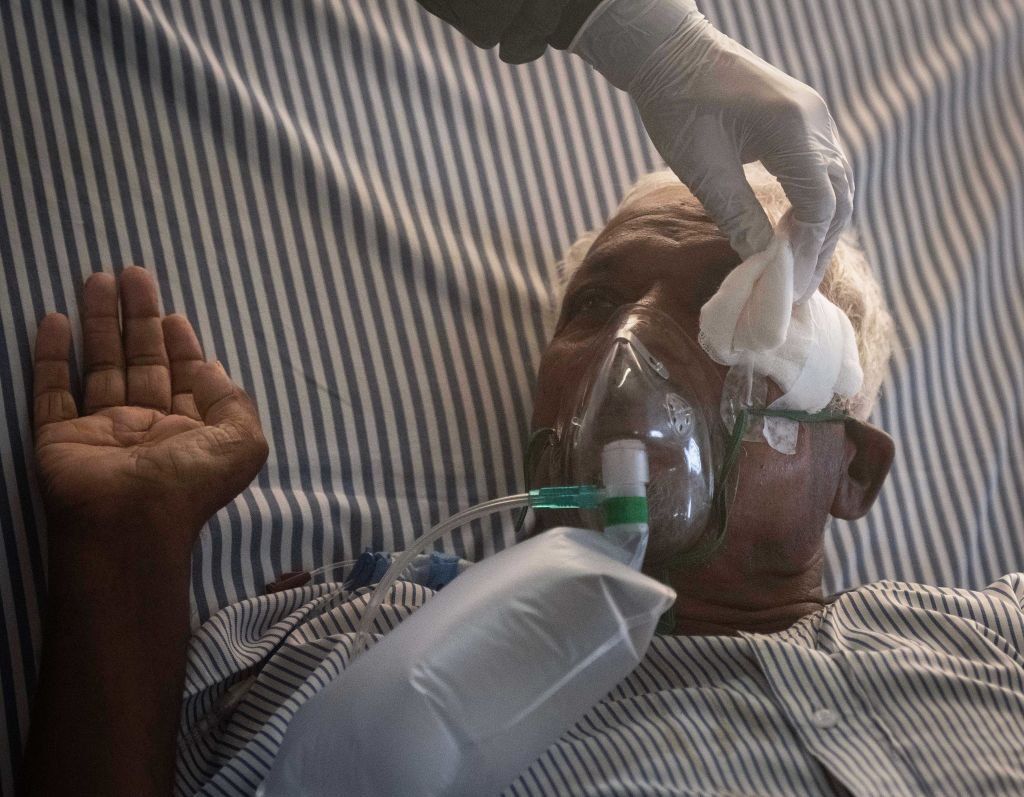
The Food and Drug Administration is on the verge of announcing one of its most contentious decisions in years: the fate of an Alzheimer’s drug that could be the first treatment approved after nearly two decades of failed efforts to find ways to curb the debilitating disease.On Monday, the agency will rule on the drug, aducanumab, which aims to slow progression of memory and thinking problems early in the disease. If approved, it would be the first new Alzheimer’s medication since 2003 and the first treatment on the market that attacks the disease process rather than just easing symptoms.It would become a blockbuster drug within several years, analysts predict, costing tens of thousands of dollars annually per patient and bringing a windfall to its manufacturer, Biogen.Patient groups, desperate for treatments, are pushing for approval. But greenlighting the drug would fly in the face of objections from several prominent Alzheimer’s experts and the F.D.A.’s independent advisory committee.In November, the committee voted overwhelmingly against recommending approval, saying data failed to demonstrate persuasively that aducanumab slowed cognitive decline. Three advisory committee members later wrote a point-by-point critique of the evidence. Other scientists, and an independent think tank, say aducanumab hadn’t shown convincing benefit to outweigh its safety risks.“This should not be approved, because substantial evidence of effectiveness hasn’t been shown,” said Dr. Lon Schneider, director of the California Alzheimer’s Disease Center at the University of Southern California and one of many site investigators who helped conduct one of the aducanumab trials. “There’s very little potential that this will address the needs of patients.”Beyond the status of this particular drug, some experts worry approval could lower standards for future drugs — an especially important question at a time when public trust in science is teetering.“I simply don’t see a path for approval because of the absence of evidence that’s been shared to date that this product works, and I think it would set a remarkably dangerous precedent — not only for the field of Alzheimer’s research but also for the broader regulation of prescription drugs in our country,” said Dr. G. Caleb Alexander, an F.D.A. advisory committee member and an internist, epidemiologist and drug safety and effectiveness expert at the Johns Hopkins Bloomberg School of Public Health.About six million people in the United States and roughly 30 million globally have Alzheimer’s, a number expected to double by 2050. Currently, five medications approved in the United States can delay cognitive decline for several months in various Alzheimer’s stages. About two million Americans have mild Alzheimer’s-related impairment, fitting criteria for aducanumab, a monthly intravenous infusion requiring regular imaging to detect potential brain swelling.Biogen officials declined to comment for this article, but in earnings calls, medical meetings and F.D.A. presentations, they have said the evidence shows cognitive benefit. Several Alzheimer’s experts whose experience includes consulting for Biogen wrote recently that aducanumab “achieves the standard of meaningful efficacy with adequate safety.”The Biogen headquarters in Cambridge, Mass.Cody O’Loughlin for The New York TimesDebate centers on two never fully completed Phase 3 trials that contradicted each other. One suggested that a high dose could slightly slow cognitive decline; the other showed no benefit. Biogen says that given the need for Alzheimer’s medications, the single positive trial, plus results from a small safety trial and aducanumab’s ability to reduce a key protein, should justify approval.The F.D.A. typically follows advisory committee recommendations and usually requires two convincing studies for approval, but it has made exceptions, especially for severe diseases that lack treatments.Two other medications now in trials appear more promising than aducanumab, experts say, but it could be three or four years before data would indicate whether they merit approval. Many families say that’s too long to wait.“There’s lots of issues with the data,” acknowledged Maria Carrillo, chief science officer for the Alzheimer’s Association, a patient advocacy group campaigning vigorously for approval. But she said her organization must “weigh the crushing reality of what people live with today” and support giving patients something to try instead of waiting several years for more conclusive positive results.The F.D.A. itself seems divided. In advisory committee presentations, a clinical analyst cited “substantial evidence of effectiveness to support approval.” But an F.D.A. statistician wrote that another trial was needed because “there is no compelling, substantial evidence of treatment effect or disease slowing.”And some experts, like Dr. Ronald Petersen, director of the Mayo Clinic’s Alzheimer’s Disease Research Center in Rochester, Minn., say they’re “on the fence.” He said he’d like to give patients a new option soon but “the data are iffy.”Aducanumab, a monoclonal antibody, targets a protein, amyloid, that clumps into plaques in the brains of Alzheimer’s patients. Many amyloid-reducing drugs failed to slow symptoms in trials, a history that, some experts say, makes it especially important that aducanumab’s data be convincing. If effective, it would support a long-held, unproven theory that attacking amyloid can help if done early enough.Excitement about aducanumab grew after a small early trial to evaluate safety showed amyloid reduction and hinted it might slow cognitive decline. The F.D.A., in a move some experts question, allowed Biogen to skip Phase 2 trials and conduct two Phase 3 trials of about 1,640 patients each.Both trials were stopped early, in March 2019, when an independent data monitoring committee said aducanumab didn’t appear to be working. Consequently, 37 percent of participants never completed the 78-week trials.But that October, Biogen announced it found benefit in one trial after evaluating data from 318 participants who finished before the trials were stopped but after the cutoff point for results the monitoring committee assessed.In that trial, Biogen said, the highest dose slowed cognitive decline by 22 percent, or about four months over 18 months. A lower dose in that trial and high and low doses in the other showed no statistically significant benefit over a placebo.“One study was positive, and one identically performed study was negative,” said Dr. David Knopman, a clinical neurologist at the Mayo Clinic and a site principal investigator for one trial. “I don’t think it takes a Ph.D. in statistics to see that that’s inconclusive.”Dewayne Nash, whose mother and brother died of Alzheimer’s, said that for his personal situation, he favored approval because he might decline before other treatments became available. But scientifically, “they really ought to do the studies” first, he said, adding that otherwise, “you’re giving people a drug that may help, but it may not.”Daniel Dreifuss for The New York TimesDr. Alexander added that Biogen’s interpretation of data using after-the-fact analyses was “like the Texas sharpshooter fallacy — the idea that the sharpshooter shoots up a barn and then goes and draws a bull’s-eye around the cluster of holes that he likes.” By contrast, Dr. Stephen Salloway, who has received research and consulting fees from Biogen but wasn’t paid for being an aducanumab trial site principal investigator, called himself a “passionate” supporter of approval. He considers the evidence sufficient because Alzheimer’s is so disabling.“I understand people’s concerns — the data set has issues, of course,” said Dr. Salloway, director of neurology and the Memory and Aging Program at Butler Hospital in Providence, R.I. “F.D.A. is in a tough spot, obviously.”But he favors giving patients the option. Of his 17 participants in both the safety trial and Phase 3, he said, 10 had remained relatively cognitively stable for several years, while seven had declined at typical rates.“It didn’t work for everybody,” he said, but “it just seemed like there were more people that were steady for longer than I’m used to.” One challenge with assessing impact is that many early-stage patients decline slowly anyway, Dr. Schneider said.Advocates and many patients say delaying deterioration even slightly is meaningful. But some experts say the single trial’s slowing of 0.39 on an 18-point scale rating memory, problem-solving skills and function may be imperceptible to patients’ experience and doesn’t justify approving a drug that floundered in another trial and carries risk of harm.“This product, even in the best of circumstances, would be not terribly effective at all, with significant safety risks,” Dr. Alexander said.The potential harm involves brain swelling or bleeding experienced by about 40 percent of Phase 3 trial participants receiving the high dose. Most were either asymptomatic or had headaches, dizziness or nausea. But such effects prompted 6 percent of high-dose recipients to discontinue. No Phase 3 participants died from the effects, but one safety trial participant did.Henry Magendantz, center, with his wife, Kathy Jellison, who believes his initial years on the drug helped slow his decline enough to allow him to help choose the assisted-living facility where he lives now. “It brought us some time,” she said. Kayana Szymczak for The New York TimesSome trial participants’ views reflect the situation’s complexity.Dewayne Nash, 71, of Santa Barbara, Calif., learned after the trial that he had received 18 months of a placebo, during which his cognitive scores improved — partly, he believes, because he lowered his cholesterol. Dr. Nash, a retired family physician, then received seven months of aducanumab, scaling up to the high dose, hoping it would slow decline, but “I didn’t notice any difference.”Dr. Nash, whose mother and brother died of Alzheimer’s, will resume aducanumab soon through Biogen’s study for previous participants. He said that for his situation, he would like it approved because he expects to decline before other therapies become available and is willing to risk “brain bleeding and stuff.”But scientifically, “I don’t like it when they rush drugs,” he said.“They really ought to do the studies that need to be done” before approval, he added. Otherwise, “you’re giving people a drug that may help, but it may not.”Dr. Salloway said one trial patient whose dementia had remained mild considerably longer than he’d expected was Henry Magendantz, a retired obstetrician-gynecologist in Providence, R.I. Dr. Magendantz, 84, started the safety trial after his wife, Kathy Jellison, noticed him having trouble following steps to assemble furniture.He received a year of placebo, then a year of lower-dose aducanumab, then two years of the high dose before the 2019 halt. During that time, Ms. Jellison said, he was “slipping a bit,” but she believes aducanumab slowed decline enough to allow him to participate in tasks like choosing an assisted-living facility, where he moved in October 2018.“It brought us some time,” she said. Debby Rosencrantz, a trial participant, hopes for Alzheimer’s treatments that work well enough to curb her dementia. “It just feels like there’s a blank in places where there shouldn’t be a blank in my brain,” she said.Kayana Szymczak for The New York TimesAnother issue with evaluating treatments is that some assessment scales, including in the aducanumab trials, involve reports from relatives or caregivers, who might miss subtle symptom progression.“It is squishy stuff,” said Susan Woskie, a professor emeritus in public health at the University of Massachusetts Lowell, whose wife, Debby Rosenkrantz, 68, participated in the trial. “This stuff is really difficult, I think, to compile into metrics that have any validity.”Ms. Rosenkrantz, a former social worker in Cambridge, Mass., said that while receiving roughly eight months of low-dose aducanumab in the trial, “I was really optimistic that there was a drug, and so for me it was like, yes, it’s working.”Since restarting infusions in Biogen’s study for previous participants last September, though, “I haven’t noticed any change,” she said.She experiences short-term memory loss and cannot follow recipes. “It just feels like there’s a blank in places where there shouldn’t be a blank in my brain,” she said.Dr. Woskie said the couple yearns for treatments but that if the F.D.A. told Biogen, “‘No, we don’t fast-track approve you; come back when you have more data,’ that wouldn’t surprise me, and it might make sense.”Some doctors who consider aducanumab’s evidence weak, including Dr. Knopman, say that if it is approved, they would tell patients their reservations but would feel ethically compelled to offer it. Still, Dr. Jason Karlawish, a co-director of the University of Pennsylvania’s Penn Memory Center and a site investigator on Biogen-sponsored studies, said, “Physicians like me, who would be prescribers, are saying, ‘I want an effective drug to prescribe to my patients — but this is not the drug.’”
Read more →