Johns Hopkins scientists grow a mini human brain that lights up and connects like the real thing
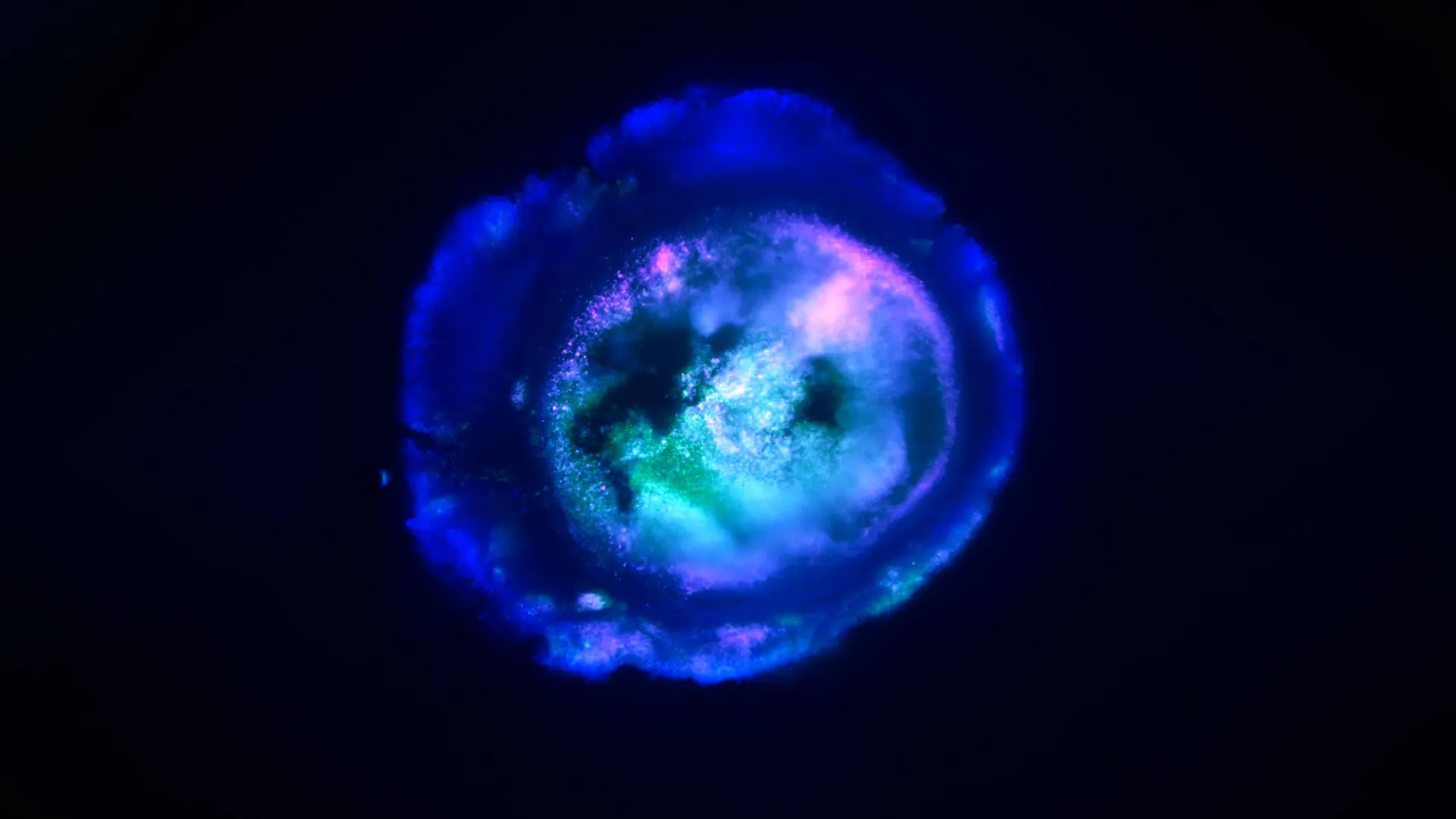
Johns Hopkins University researchers have grown a novel whole-brain organoid, complete with neural tissues and rudimentary blood vessels — an advance that could usher in a new era of research into neuropsychiatric disorders such as autism.
“We’ve made the next generation of brain organoids,” said lead author Annie Kathuria, an assistant professor in JHU’s Department of Biomedical Engineering who studies brain development and neuropsychiatric disorders. “Most brain organoids that you see in papers are one brain region, like the cortex or the hindbrain or midbrain. We’ve grown a rudimentary whole-brain organoid; we call it the multi-region brain organoid (MRBO).”
The research, published in Advanced Science, marks one of the first times scientists have been able to generate an organoid with tissues from each region of the brain connected and acting in concert. Having a human cell-based model of the brain will open possibilities for studying schizophrenia, autism, and other neurological diseases that affect the whole brain — work that typically is conducted in animal models.
To generate a whole-brain organoid, Kathuria and members of her team first grew neural cells from the separate regions of the brain and rudimentary forms of blood vessels in separate lab dishes. The researchers then stuck the individual parts together with sticky proteins that act as a biological superglue and allowed the tissues to form connections. As the tissues began to grow together, they started producing electrical activity and responding as a network.
The multi-region mini brain organoid retained a broad range of types of neuronal cells, with characteristics resembling a brain in a 40-day-old human fetus. Some 80% of the range of types of cells normally seen at the early stages of human brain development was equally expressed in the laboratory-crafted miniaturized brains. Much smaller compared to a real brain — weighing in at 6 million to 7 million neurons compared with tens of billions in adult brains — these organoids provide a unique platform on which to study whole-brain development.
The researchers also saw the creation of an early blood-brain barrier formation, a layer of cells that surround the brain and control which molecules can pass through.
“We need to study models with human cells if you want to understand neurodevelopmental disorders or neuropsychiatric disorders, but I can’t ask a person to let me take a peek at their brain just to study autism,” Kathuria said. “Whole-brain organoids let us watch disorders develop in real time, see if treatments work, and even tailor therapies to individual patients.”
Using whole-brain organoids to test experimental drugs may also help improve the rate of clinical trial success, researchers said. Roughly 85% to 90% of drugs fail during Phase 1 clinical trials. For neuropsychiatric drugs, the fail rate is closer to 96%. This is because scientists predominantly study animal models during the early stages of drug development. Whole-brain organoids more closely resemble the natural development of a human brain and likely will make better test subjects.
“Diseases such as schizophrenia, autism, and Alzheimer’s affect the whole brain, not just one part of the brain. If you can understand what goes wrong early in development, we may be able to find new targets for drug screening,” Kathuria said. “We can test new drugs or treatments on the organoids and determine whether they’re actually having an impact on the organoids.”