Ancient DNA reveals leprosy hit the Americas long before colonization
Hansen’s Disease (leprosy) is thought to have originated in Eurasia: previous studies on Mycobacterium leprae, the dominant form of leprosy, suggest the disease originated in Eurasia. Ancient pathogen genomes from old bones: a team of scientists from Germany and Argentina have reconstructed two genomes of Mycobacterium lepromatosis in 4000-year-old human skeletons from Chile. This pathogen is regarded as a second, less common, cause of Hansen’s Disease. A new American chapter for Hansen’s Disease: two pathogens that are responsible for the same disease evolved separately on opposite sides of the world for thousands of years.Hansen’s Disease, more commonly known as leprosy, is a chronic disease that can lead to physical impairment. Today it exists in over 100 countries, and while the infection is treatable, access to treatment varies widely with socioeconomic conditions. Its mention in historical texts give us a glimpse into its past impact on population health in Europe and Asia. Prolonged untreated infection can result in characteristic changes in bone, and these have been documented in archaeological skeletons as early as 5000 years ago in Europe, Asia, and Oceania. So far, absence of these characteristic changes in the pre-contact American contexts suggests that leprosy was one of the many diseases introduced to the continent in the colonial period. Thereafter it afflicted humans and curiously also armadillos.From a genetic perspective, Hansen’s Disease is caused by either the globally dominant Mycobacterium leprae or the newly identified and rare Mycobacterium lepromatosis. The recovery of M. leprae from archaeological bone in Europe suggests the disease originated in Eurasia sometime during the Neolithic transition about 7000 years ago. Similar emergence estimates have been proposed for other notorious diseases such as plague, tuberculosis, and typhoid fever. Ancient genomes for M. lepromatosis have remained elusive, and these may hold important clues on the history of Hansen’s Disease.
Past disease in the American continent
We know comparatively little about the infectious disease experience of the diverse communities of people living in the Americas before the colonial period. This accounts for almost 20,000 years of human history, and the diverse ecosystems into which humans integrated across the continent would have presented challenges to the immune system not otherwise encountered in other parts of the world. We know very little of these diseases, as they were smothered by the onslaught of pathogens that Europeans later brought with them. Archaeological study of human bones from the pre-contact Americas confirm that the time was far from disease-free, but often the traces seen on the bones aren’t specific enough to assign to a known disease.
“Ancient DNA has become a great tool that allows us to dig deeper into diseases that have had a long history in the Americas,” says Kirsten Bos, group leader for Molecular Paleopathology at the Max Planck Institute for Evolutionary Anthropology. She and her team have been studying pathological bone from American context for over a decade. Some diseases that the group has found were expected – just last year they found evidence that the family of diseases closely related to syphilis had its roots in the Americas, which many had suspected. “The advanced techniques now used to study ancient pathogen DNA allows us to look beyond the suspects and into other diseases that might not be expected from the context,” she adds.
Re-writing the history of Hansen’s Disease
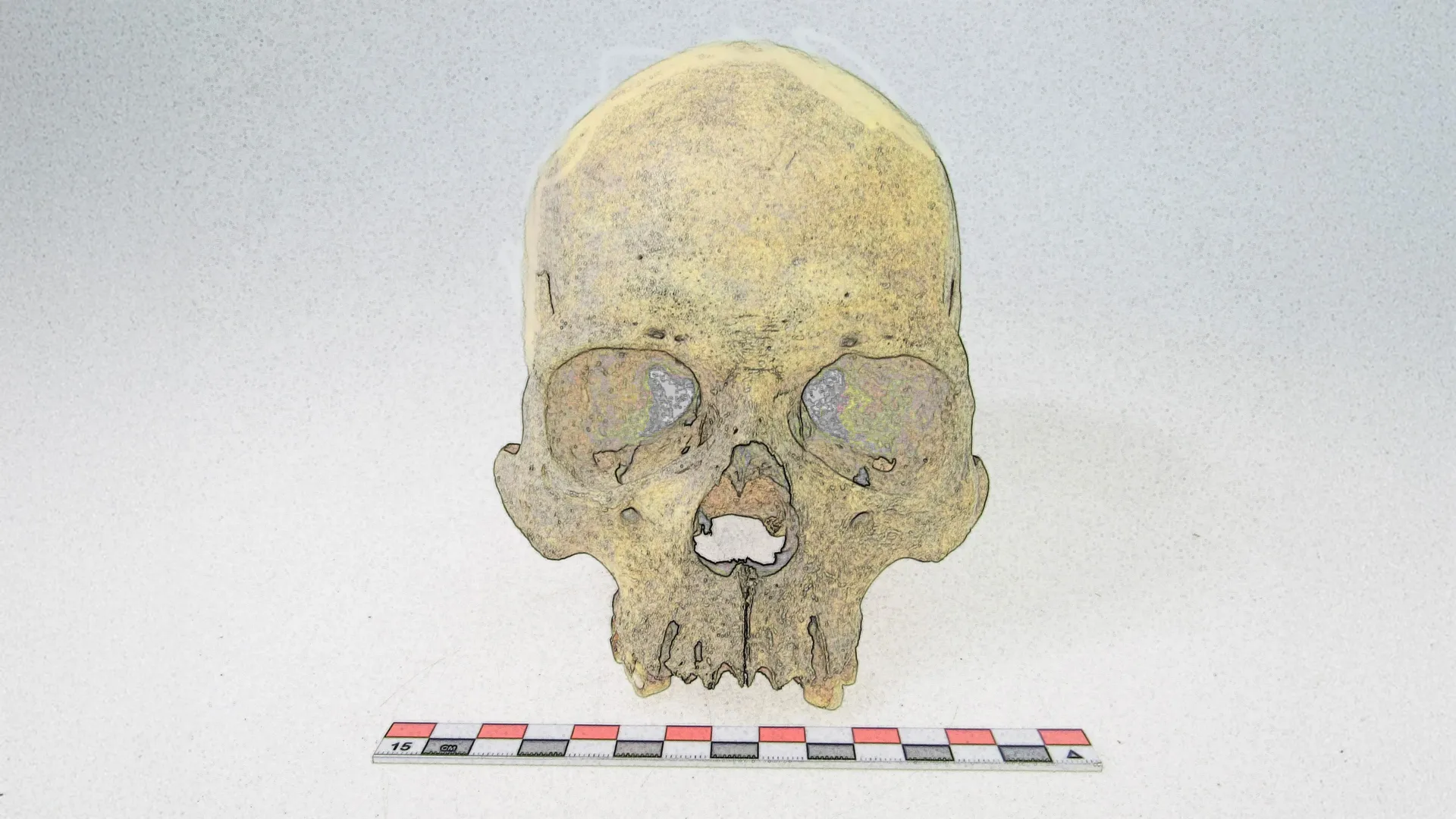
Bos’s team worked closely with researchers from Argentina and Chile to both identify bones suitable for analysis and to carry out the meticulous work of isolating the DNA of ancient pathogens. Doctoral candidate Darío Ramirez of the University of Córdoba, Argentina, worked extensively with such material, and was the first to identify a genetic signature related to leprosy in some 4000-year-old skeletons from Chile. “We were initially suspicious, since leprosy is regarded a colonial-era disease, but more careful evaluation of the DNA revealed the pathogen to be of the lepromatosis form.” This provided the first clue that M. lepromatosis and M. leprae, though nominally both pathogens that cause Hansen’s Disease, might have very different histories. Reconstruction of the genome was key in looking into this. While putting the molecular puzzle of an ancient genome back together is never an easy task, these pathogens in particular had “amazing preservation, which is uncommon in ancient DNA, especially from specimens of that age,” comments Lesley Sitter, a postdoctoral researcher in Bos’s team who carried out the analysis.
The pathogen is related to all known modern forms of M. lepromatosis, but as there are so few genomes available for comparison, there is still much to be learned about it. This work has shown that a pathogen considered rare in a modern context caused disease for thousands of years in the Americas. Rodrigo Nores, professor of Anthropology at the University of Córdoba, Argentina is convinced that more cases, both ancient and modern, will be identified in the coming years: “this disease was present in Chile as early as 4000 years ago, and now that we know it was there, we can specifically look for it in other contexts.” Once more genomes surface, we’ll be able to piece out further details of its history and better understand its global distribution today. The pathogen has recently been discovered in squirrel populations from the United Kingdom and Ireland, but in the Americas it has yet to be found in any species other than humans. With such little data, mystery still surrounds its origin. “It remains to be determined if the disease originated in the Americas, or if it joined some of the first settlers from Eurasia,” adds Bos. “So far the evidence points in the direction of an American origin, but we’ll need more genomes from other time periods and contexts to be sure.”