The UK’s pioneering licensed IVF technique to reduce the risk of mitochondrial diseases carried out in Newcastle has seen eight babies born, published research shows.
All eight babies show no signs of having mitochondrial DNA disease. The babies, four girls and four boys, including one set of identical twins, were born to seven women at high risk of transmitting serious disease caused by mutations in mitochondrial DNA. The findings, reported on July 16 by the Newcastle team who pioneered mitochondrial donation using fertilized human eggs, indicate that the new treatment, known as pronuclear transfer, is effective in reducing the risk of otherwise incurable mitochondrial DNA diseases.
Published in two papers in The New England Journal of Medicine (NEJM), the findings describe the reproductive and clinical outcomes of pronuclear transfer treatments performed to date. All babies were healthy at birth, meeting their developmental milestones, and the mother’s disease-causing mitochondrial DNA mutations were either undetectable or present at levels that are very unlikely to cause disease.
The technique was pioneered in human eggs by a team based at Newcastle University, UK and the Newcastle upon Tyne Hospitals NHS Foundation Trust in work funded by Wellcome and NHS England.
The mother of a baby girl born following mitochondrial donation said: “As parents, all we ever wanted was to give our child a healthy start in life. Mitochondrial donation IVF made that possible. After years of uncertainty this treatment gave us hope—and then it gave us our baby. We look at them now, full of life and possibility, and we’re overwhelmed with gratitude. Science gave us a chance.”
The mother of a baby boy added: “We are now proud parents to a healthy baby—a true mitochondrial replacement success. This breakthrough has lifted the heavy cloud of fear that once loomed over us.
“Thanks to this incredible advancement and the support we received, our little family is complete. The emotional burden of mitochondrial disease has been lifted, and in its place is hope, joy, and deep gratitude.”
The NHS Mitochondrial Reproductive Care Pathway offers mitochondrial donation, through a research study, in addition to other reproductive options for women with mitochondrial disease.
Professor Sir Doug Turnbull, Newcastle University part of the Newcastle team said: “Mitochondrial disease can have a devastating impact on families. Today’s news offers fresh hope to many more women at risk of passing on this condition who now have the chance to have children growing up without this terrible disease. Within the framework of the NHS in a well-regulated environment, we are able to offer mitochondrial donation as part of a research study to affected women in the UK.“
Mitochondrial DNA disease
Every year, around one in 5,000 children is born with mitochondrial DNA mutations that can cause devastating disease. Mitochondria produce the energy required for life and contain a small piece of DNA that only encodes some of the instructions required for energy production. Harmful mutations in mitochondrial DNA can result in reduced availability of energy, particularly affecting tissues that have high energy demands – for example heart, muscle and brain. Mitochondrial DNA is maternally inherited, and these diseases are therefore passed from mother to child. Although males can be affected, they do not pass on the disease. Despite years of research there is still no cure for people with mitochondrial DNA disease.
In the absence of a cure for mitochondrial DNA diseases, attention has focused on IVF-based technologies to reduce the risk of disease by limiting transmission of disease-causing mitochondrial DNA mutations from mother to child. The new IVF-based mitochondrial donation technology, pronuclear transfer, which was legalized in the UK in 2015, is designed to reduce the risk of mitochondrial DNA disease in children born to women who carry high levels of disease-causing mitochondrial DNA mutations. The Newcastle team now include pronuclear transfer as part of a research study along with a range of reproductive options offered to women at risk of transmitting mitochondrial disease to their children.
Pronuclear transfer
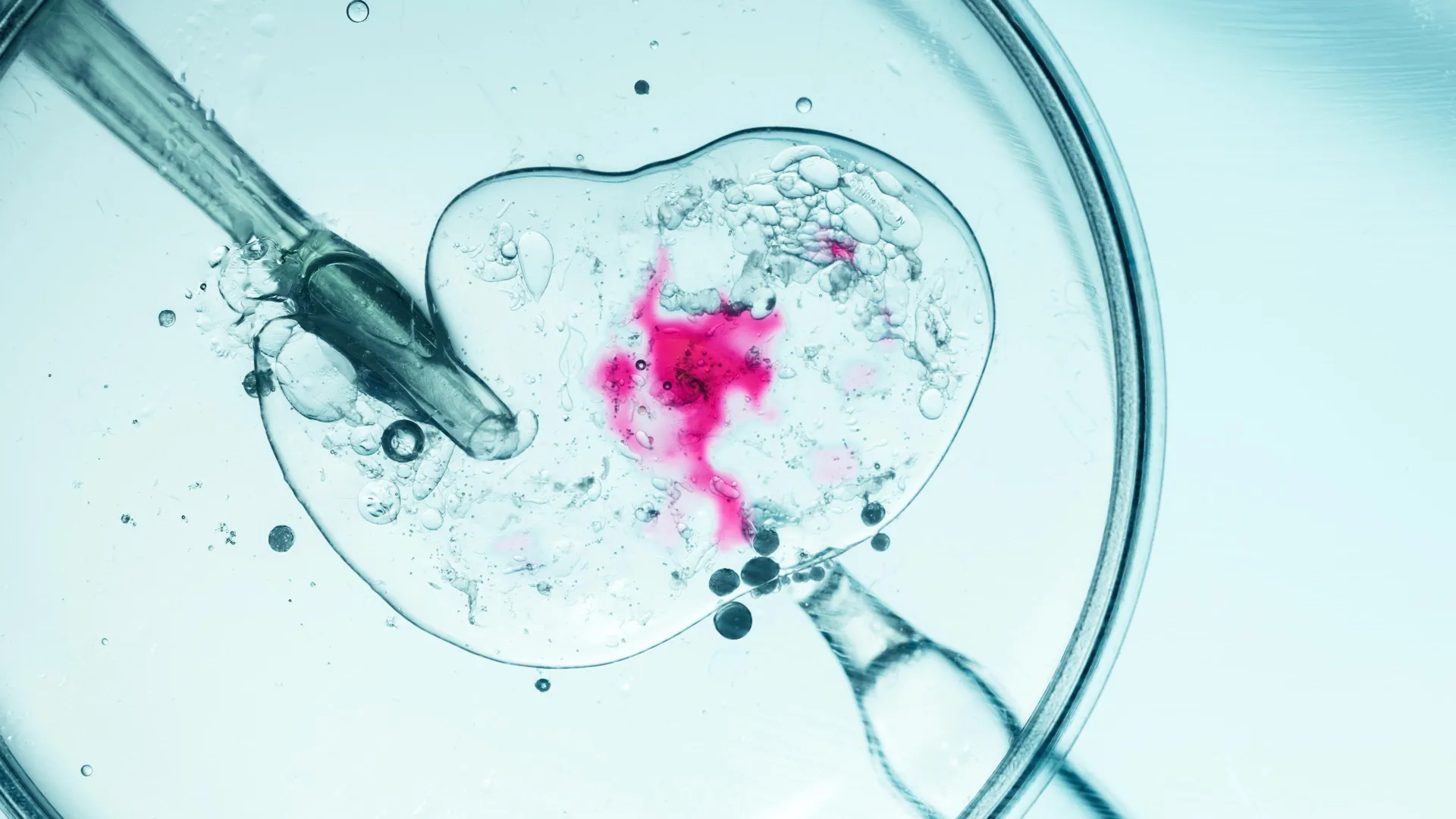
The technique, known as pronuclear transfer is performed after the egg is fertilized. It involves transplanting the nuclear genome (which contains all the genes essential for our individual characteristics, for example, hair color and height) from an egg carrying a mitochondrial DNA mutation to an egg donated by an unaffected woman that has had its nuclear genome removed. The resulting embryo inherits its parents’ nuclear DNA, but the mitochondrial DNA is inherited predominantly from the donated egg.
The reproductive outcomes paper
The UK-based Newcastle team who developed and optimized pronuclear transfer for use in fertilized human eggs now report on outcomes of pronuclear-transfer treatment to reduce the risk of mitochondrial DNA disease.
Levels of disease-causing mitochondrial DNA detected in babies born after pronuclear transfer treatment ranged from undetectable to 16% in neonatal blood. The presence of mitochondrial DNA mutations in babies born after pronuclear transfer treatment results from carryover of maternal mitochondria surrounding the nuclear DNA at the time of transplantation. Carryover of maternal mitochondrial DNA is a known limitation of mitochondrial donation technologies.
The team is seeking to better understand and address this issue as part of an underpinning research program.
Professor Mary Herbert, lead author of the reproductive outcomes paper who carried out the research at Newcastle University said: “The findings give grounds for optimism. However, research to better understand the limitations of mitochondrial donation technologies, will be essential to further improve treatment outcomes.
“Mitochondrial donation technologies are currently regarded as risk reduction treatments owing to carryover of maternal mitochondrial DNA during the mitochondrial donation procedure. Our ongoing research seeks to bridge the gap between risk reduction and prevention of mitochondrial DNA disease by addressing this problem.”
Pronuclear-transfer treatment is offered as part of an integrated program that includes preimplantation genetic testing (PGT) for reducing the risk of mitochondrial DNA disease. In accordance with HFEA regulations, pronuclear transfer is offered only to those women who are unlikely to benefit from PGT treatment.At the time of reporting the integrated program of PGT and pronuclear transfer, clinical pregnancies were confirmed in 8 of 22 (36%) patients who underwent pronuclear transfer and 16 of 39 (41%) of patients who underwent PGT. Pronuclear transfer has resulted in eight births and one further pregnancy. PGT has resulted in 18 births. In the children from pronuclear transfer, levels of disease-causing mitochondrial DNA mutations were either undetectable or well below the levels at which disease symptoms are observed.
The clinical outcomes paper
The Newcastle team describe the pathway developed to provide the best possible care for women with pathogenic mitochondrial DNA mutations. It describes in detail how the mothers of the first children born with the technique were monitored and supported in pregnancy, and their babies closely followed from birth.
Some of the mothers already had symptoms of mitochondrial disease including vision loss and heart problems. Others had family members with the disease and remain at risk of developing symptoms and passing it on.
All eight babies, including a set of identical twins, were healthy at birth and are described as developing normally – five have had no medical problems since. In the paper, the team note that three babies overcame some early health issues that they believe they are not able to attribute directly to mitochondrial donation.
The Newcastle team offers advice and treatment to women with harmful mitochondrial DNA mutations in the UK. They are carefully monitored during pregnancy and after mitochondrial donation, six of seven progressed without incident. One woman developed a rare complication of pregnancy with a high level of fats detected in her blood (hyperlipidaemia) which responded well to a reduced fat diet.
All eight babies, including the set of twins, were born by normal vaginal delivery or elective caesarean section. All babies had normal weight for gestational age. The level of disease-causing mitochondrial DNA mutation was measured in blood and urine cells and was undetectable in five babies. Three babies had low levels of disease-causing mitochondrial DNA mutations – 5 and 9%, 12 and 13%, 16 and 20% in blood and urine respectively. These levels are well below the 80% level required for clinical disease for these mutations. The researchers note that at follow-up at 18 months, the level of the disease-causing mutation in the child with 5 and 9% was undetectable in blood and urine.
All children are enrolled in an 18-month developmental study and at the date of reporting all the babies were meeting their relevant developmental milestones.One child developed some brief startles (involving neck flexion and eye blinking) at age 7 months, which resolved without treatment after 3 months. Another, a breast-fed baby, developed high blood fats (hyperlipidaemia) which had also affected the mother during pregnancy, and was successfully treated through a low-fat diet. This child was also diagnosed with an abnormal heart rhythm (cardiac arrhythmia) which is being successfully treated with a reducing amount of anti-arrhythmic medication. (Although the children born following PGT are not routinely followed-up, the team note that a cardiac anomaly was detected in one child.) A third child had a urinary tract infection that responded quickly to antibiotic treatment.
The authors say that the children’s health conditions are not thought to be related to the maternal mitochondrial DNA mutations as the low levels detected in these babies would not be expected to cause disease symptoms. Symptoms for these mutations are only seen with levels above 80%. Any effect of the pronuclear transfer procedure itself would be expected to have a more uniform clinical manifestation, that is, to affect children in the same way. However, follow-up studies will be of paramount importance in detecting any patterns in childhood conditions.
The team emphasize that follow-up studies are essential for detecting any patterns in childhood conditions and say they will continue to offer assessments up to the age of 5 years.
Professor Bobby McFarland, Director of the NHS Highly Specialised Service for Rare Mitochondrial Disorders (Newcastle Hospitals NHS Foundation Trust) and Professor of Paediatric Mitochondrial Medicine at Newcastle University is first author of one of the papers. He said: “While longer term follow-up of children born following mitochondrial donation is of paramount importance, these early results are very encouraging. Seeing the joy and relief these children have brought to their parents is such a privilege.
“We believe the follow-up process we have put in place is thorough, since it allows us to detect and review even minor health conditions in children born after pronuclear transfer such as a urinary tract infection.”
The Lily Foundation, a charity dedicated to fighting mitochondrial disease has supported the Newcastle work. “We’re absolutely delighted with the results of these published papers,” said Liz Curtis, Lily founder and CEO. “We fought long and hard for this change so that families could have choices. After years of waiting, we now know that eight babies have been born using this technique, all showing no signs of mito. For many affected families, it’s the first real hope of breaking the cycle of this inherited condition.”
FACT FILE
Law – In a first worldwide and following extensive public debate and scientific and ethical review, UK legalization was press/articles/archive/2015/10/worldfirstledbynewcastleuniversity/” >approved in 2015 to enable the Human Fertilisation and Embryology Authority (HFEA) to allow mitochondrial donation treatments for women at high risk of transmitting serious mitochondrial DNA disease to their children. Following this, the law has now changed in Australia.
Licence – Licences are regulated and granted by the HFEA. Newcastle Fertility Centre part of Newcastle Hospitals NHS Foundation Trust was granted the press/articles/archive/2017/03/mitochondrialicence/” >first license to perform clinical mitochondrial donation by pronuclear transfer in 2017. A clinical pathway was established with mitochondrial clinicians as part of NHS England’s Highly Specialised Service.
Mitochondrial disease refers to a group of genetic conditions that disrupt how our mitochondria – the energy producers in our cells – function.
Pre-implantation genetic testing (PGT) is a procedure that helps couples avoid passing on genetic conditions to their children. This extra step tests embryos for genetic conditions.
Pronuclear transfer (PNT) involves transferring the nuclear DNA of a fertilized egg into a fertilized donor egg to prevent the transmission of mitochondrial DNA (mtDNA) disease.
Funding
The team acknowledge that the Mitochondrial Reproductive Care Pathway is supported by the NHS at The Newcastle upon Tyne Hospitals NHS Foundation Trust (NUTH). Support was provided by Wellcome. Infrastructural support was provided by Newcastle University, a National Institute for Health and Care Research (NIHR) Biomedical Research Centre award to NUTH. The NHS Highly Specialised Services for Rare Mitochondrial Disorders is supported by NHS England and a career development award was made to Dr Hyslop from Health Education England and the NIHR.
Read more →