Researchers Are Hatching a Low-Cost Coronavirus Vaccine
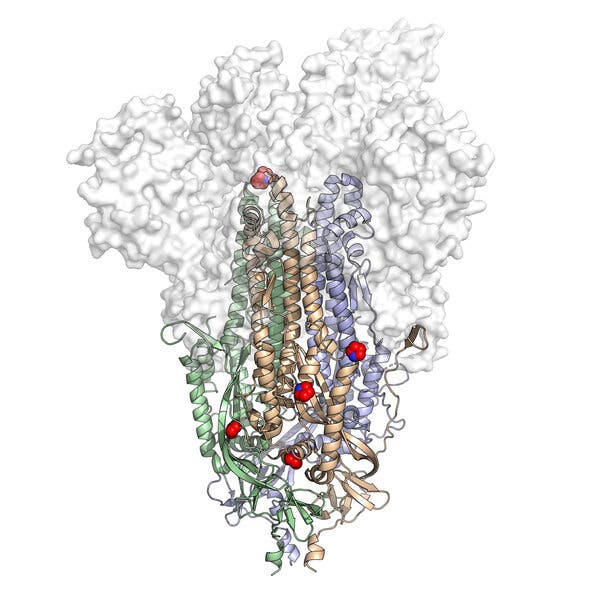
A new formulation entering clinical trials in Brazil, Mexico, Thailand and Vietnam could change how the world fights the pandemic.A new vaccine for Covid-19 that is entering clinical trials in Brazil, Mexico, Thailand and Vietnam could change how the world fights the pandemic. The vaccine, called NVD-HXP-S, is the first in clinical trials to use a new molecular design that is widely expected to create more potent antibodies than the current generation of vaccines. And the new vaccine could be far easier to make.Existing vaccines from companies like Pfizer and Johnson & Johnson must be produced in specialized factories using hard-to-acquire ingredients. In contrast, the new vaccine can be mass-produced in chicken eggs — the same eggs that produce billions of influenza vaccines every year in factories around the world.If NVD-HXP-S proves safe and effective, flu vaccine manufacturers could potentially produce well over a billion doses of it a year. Low- and middle-income countries currently struggling to obtain vaccines from wealthier countries may be able to make NVD-HXP-S for themselves or acquire it at low cost from neighbors.“That’s staggering — it would be a game-changer,” said Andrea Taylor, assistant director of the Duke Global Health Innovation Center.First, however, clinical trials must establish that NVD-HXP-S actually works in people. The first phase of clinical trials will conclude in July, and the final phase will take several months more. But experiments with vaccinated animals have raised hopes for the vaccine’s prospects.“It’s a home run for protection,” said Dr. Bruce Innes of the PATH Center for Vaccine Innovation and Access, which has coordinated the development of NVD-HXP-S. “I think it’s a world-class vaccine.”2P to the rescueThe molecular structure of HexaPro, a modified version of the SARS-CoV-2 spike protein, with its six key alterations shown as red and blue spheres.University of Texas at AustinVaccines work by acquainting the immune system with a virus well enough to prompt a defense against it. Some vaccines contain entire viruses that have been killed; others contain just a single protein from the virus. Still others contain genetic instructions that our cells can use to make the viral protein.Once exposed to a virus, or part of it, the immune system can learn to make antibodies that attack it. Immune cells can also learn to recognize infected cells and destroy them.In the case of the coronavirus, the best target for the immune system is the protein that covers its surface like a crown. The protein, known as spike, latches onto cells and then allows the virus to fuse to them.But simply injecting coronavirus spike proteins into people is not the best way to vaccinate them. That’s because spike proteins sometimes assume the wrong shape, and prompt the immune system to make the wrong antibodies.Jason McLellan, a structural biologist at the University of Texas at Austin. His research on coronavirus spike proteins aided the development of the Pfizer, Moderna, Johnson & Johnson and Novavax vaccines.Ilana Panich-Linsman for The New York TimesThis insight emerged long before the Covid-19 pandemic. In 2015, another coronavirus appeared, causing a deadly form of pneumonia called MERS. Jason McLellan, a structural biologist then at the Geisel School of Medicine at Dartmouth, and his colleagues set out to make a vaccine against it.They wanted to use the spike protein as a target. But they had to reckon with the fact that the spike protein is a shape-shifter. As the protein prepares to fuse to a cell, it contorts from a tulip-like shape into something more akin to a javelin.Scientists call these two shapes the prefusion and postfusion forms of the spike. Antibodies against the prefusion shape work powerfully against the coronavirus, but postfusion antibodies don’t stop it.Dr. McLellan and his colleagues used standard techniques to make a MERS vaccine but ended up with a lot of postfusion spikes, useless for their purposes. Then they discovered a way to keep the protein locked in a tulip-like prefusion shape. All they had to do was change two of more than 1,000 building blocks in the protein into a compound called proline.The resulting spike — called 2P, for the two new proline molecules it contained — was far more likely to assume the desired tulip shape. The researchers injected the 2P spikes into mice and found that the animals could easily fight off infections of the MERS coronavirus.The team filed a patent for its modified spike, but the world took little notice of the invention. MERS, although deadly, is not very contagious and proved to be a relatively minor threat; fewer than 1,000 people have died of MERS since it first emerged in humans.But in late 2019 a new coronavirus, SARS-CoV-2, emerged and began ravaging the world. Dr. McLellan and his colleagues swung into action, designing a 2P spike unique to SARS-CoV-2. In a matter of days, Moderna used that information to design a vaccine for Covid-19; it contained a genetic molecule called RNA with the instructions for making the 2P spike.Other companies soon followed suit, adopting 2P spikes for their own vaccine designs and starting clinical trials. All three of the vaccines that have been authorized so far in the United States — from Johnson & Johnson, Moderna and Pfizer-BioNTech — use the 2P spike.Other vaccine makers are using it as well. Novavax has had strong results with the 2P spike in clinical trials and is expected to apply to the Food and Drug Administration for emergency use authorization in the next few weeks. Sanofi is also testing a 2P spike vaccine and expects to finish clinical trials later this year.Two prolines are good; six are betterDr. McLellan’s ability to find lifesaving clues in the structure of proteins has earned him deep admiration in the vaccine world. “This guy is a genius,” said Harry Kleanthous, a senior program officer at the Bill & Melinda Gates Foundation. “He should be proud of this huge thing he’s done for humanity.”But once Dr. McLellan and his colleagues handed off the 2P spike to vaccine makers, he turned back to the protein for a closer look. If swapping just two prolines improved a vaccine, surely additional tweaks could improve it even more.“It made sense to try to have a better vaccine,” said Dr. McLellan, who is now an associate professor at the University of Texas at Austin.In March, he joined forces with two fellow University of Texas biologists, Ilya Finkelstein and Jennifer Maynard. Their three labs created 100 new spikes, each with an altered building block. With funding from the Gates Foundation, they tested each one and then combined the promising changes in new spikes. Eventually, they created a single protein that met their aspirations.The winner contained the two prolines in the 2P spike, plus four additional prolines found elsewhere in the protein. Dr. McLellan called the new spike HexaPro, in honor of its total of six prolines.The structure of HexaPro was even more stable than 2P, the team found. It was also resilient, better able to withstand heat and damaging chemicals. Dr. McLellan hoped that its rugged design would make it potent in a vaccine.Dr. McLellan also hoped that HexaPro-based vaccines would reach more of the world — especially low- and middle-income countries, which so far have received only a fraction of the total distribution of first-wave vaccines.“The share of the vaccines they’ve received so far is terrible,” Dr. McLellan said.To that end, the University of Texas set up a licensing arrangement for HexaPro that allows companies and labs in 80 low- and middle-income countries to use the protein in their vaccines without paying royalties.Meanwhile, Dr. Innes and his colleagues at PATH were looking for a way to increase the production of Covid-19 vaccines. They wanted a vaccine that less wealthy nations could make on their own.With a little help from eggsThe first wave of authorized Covid-19 vaccines require specialized, costly ingredients to make. Moderna’s RNA-based vaccine, for instance, needs genetic building blocks called nucleotides, as well as a custom-made fatty acid to build a bubble around them. Those ingredients must be assembled into vaccines in purpose-built factories.The way influenza vaccines are made is a study in contrast. Many countries have huge factories for making cheap flu shots, with influenza viruses injected into chicken eggs. The eggs produce an abundance of new copies of the viruses. Factory workers then extract the viruses, weaken or kill them and then put them into vaccines.The PATH team wondered if scientists could make a Covid-19 vaccine that could be grown cheaply in chicken eggs. That way, the same factories that make flu shots could make Covid-19 shots as well.In New York, a team of scientists at the Icahn School of Medicine at Mount Sinai knew how to make just such a vaccine, using a bird virus called Newcastle disease virus that is harmless in humans.For years, scientists had been experimenting with Newcastle disease virus to create vaccines for a range of diseases. To develop an Ebola vaccine, for example, researchers added an Ebola gene to the Newcastle disease virus’s own set of genes.The scientists then inserted the engineered virus into chicken eggs. Because it is a bird virus, it multiplied quickly in the eggs. The researchers ended up with Newcastle disease viruses coated with Ebola proteins.At Mount Sinai, the researchers set out to do the same thing, using coronavirus spike proteins instead of Ebola proteins. When they learned about Dr. McLellan’s new HexaPro version, they added that to the Newcastle disease viruses. The viruses bristled with spike proteins, many of which had the desired prefusion shape. In a nod to both the Newcastle disease virus and the HexaPro spike, they called it NDV-HXP-S.PATH arranged for thousands of doses of NDV-HXP-S to be produced in a Vietnamese factory that normally makes influenza vaccines in chicken eggs. In October, the factory sent the vaccines to New York to be tested. The Mount Sinai researchers found that NDV-HXP-S conferred powerful protection in mice and hamsters.“I can honestly say I can protect every hamster, every mouse in the world against SARS-CoV-2,” Dr. Peter Palese, the leader of the research, said. “But the jury’s still out about what it does in humans.”The potency of the vaccine brought an extra benefit: The researchers needed fewer viruses for an effective dose. A single egg may yield five to 10 doses of NDV-HXP-S, compared to one or two doses of influenza vaccines.“We are very excited about this, because we think it’s a way of making a cheap vaccine,” Dr. Palese said.A nurse administering the NDV-HXP-S vaccine to a volunteer at Mahidol University in Bangkok during the country’s first human trial.Government Pharmaceutical Organization of Thailand, via Agence France-Presse — Getty ImagesPATH then connected the Mount Sinai team with influenza vaccine makers. On March 15, Vietnam’s Institute of Vaccines and Medical Biologicals announced the start of a clinical trial of NDV-HXP-S. A week later, Thailand’s Government Pharmaceutical Organization followed suit. On March 26, Brazil’s Butantan Institute said it would ask for authorization to begin its own clinical trials of NDV-HXP-S.Meanwhile, the Mount Sinai team has also licensed the vaccine to the Mexican vaccine maker Avi-Mex as an intranasal spray. The company will start clinical trials to see if the vaccine is even more potent in that form.To the nations involved, the prospect of making the vaccines entirely on their own was appealing. “This vaccine production is produced by Thai people for Thai people,” Thailand’s health minister, Anutin Charnvirakul, said at the announcement in Bangkok.From left, Dimas Covas, director of the Butantan Institute in Brazil; João Doria, governor of the state of São Paulo; and Jean Gorinchteyn, the state health secretary, announcing the ButanVac Covid-19 vaccine candidate against in São Paulo on March 26. Miguel Schincariol/Agence France-Presse — Getty ImagesIn Brazil, the Butantan Institute trumpeted its version of NDV-HXP-S as “the Brazilian vaccine,” one that would be “produced entirely in Brazil, without depending on imports.”Ms. Taylor, of the Duke Global Health Innovation Center, was sympathetic. “I could understand why that would really be such an attractive prospect,” she said. “They’ve been at the mercy of global supply chains.”Madhavi Sunder, an expert on intellectual property at Georgetown Law School, cautioned that NDV-HXP-S would not immediately help countries like Brazil as they grappled with the current wave of Covid-19 infections. “We’re not talking 16 billion doses in 2020,” she said.Instead, the strategy will be important for long-term vaccine production — not just for Covid-19 but for other pandemics that may come in the future. “It sounds super promising,” she said.In the meantime, Dr. McLellan has returned to the molecular drawing board to try to make a third version of their spike that is even better than HexaPro.“There’s really no end to this process,” he said. “The number of permutations is almost infinite. At some point, you’d have to say, ‘This is the next generation.’”
Read more →